Gamp categories examples8/13/2023 
There is also encouragement to use tools where possible to increase validation efficiency and defect detection.įor example, previous approaches to documenting the execution of a validation test case may have required the tester to initial and date each step. System information and test evidence still need to be captured but the format of the information captured can be flexible as long as it is attributable, protected against unauthorised changes and available when needed. The GAMP 5 Second Edition guide emphasises that focus should be on value-adding activities rather than documentation for documentation’s sake. The GAMP 5 Second Edition guide emphasises that focus should be on value-adding activities rather than documentation for documentation’s sake.” While that model remains relevant from a planning and workflow perspective, ISPE GAMP 5 Second Edition is fully supportive of agile development and iterative testing methodologies and practices. The validation of commercially available software applications has largely followed the GAMP V-model. Testing of Computerized Systems – Appendix D5 Taken together these appendices provide the validation practitioner with broad contemporary thinking that can be applied to their day-to-day project activities. This article primarily discusses Appendix D5 Testing of Computerized Systems and Appendix M12 Critical Thinking. Significant updates to three of the original appendices. 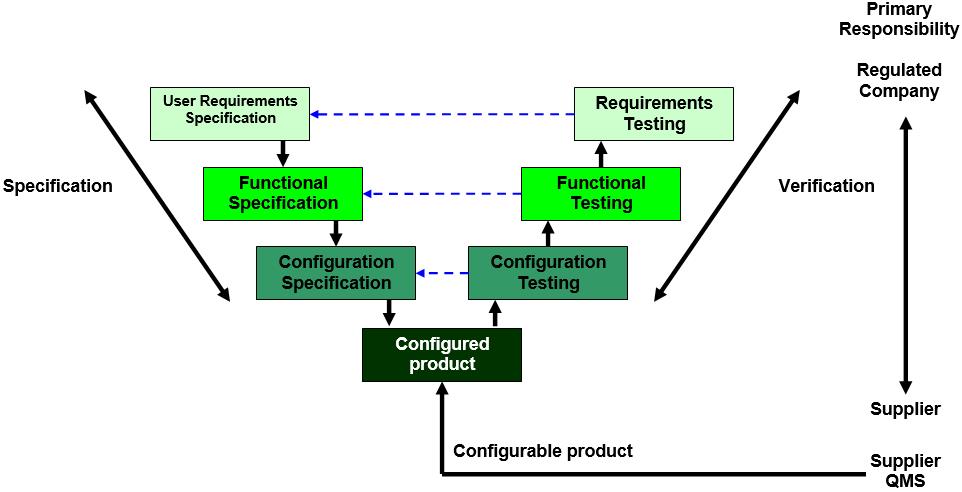
Moving from ‘snapshot’-type qualification activities to real-time monitoring and control for the IT infrastructure supports this flexibility.īalancing regulations for weighing in a GMP quality control laboratoryįor the purposes of this discussion, we will focus on what we consider to be the most significant changes to GAMP 5 but we would encourage the reader to review the guidance in its entirety within the context of their regulated business activities. This required a paradigm shift from the previous ‘validate the system and keep it static and unchanged for as long as possible’ mindset to a dynamic and flexible approach to maintaining the system in a validated state of control throughout its operational life. While much software development now follows a more agile approach, changes – in the form of updates to the operating environment and software applications – are now frequent and essential to sustain data integrity and data security as well as operational efficiency. The original GAMP core principles are now applied more broadly by the Second Edition to allow contemporary and competent practitioners the freedom to employ innovative and evolving technologies without the previously assumed documentation burden. Given the rapid acceptance of ‘as-a-service’ software models and significant outsourcing of previously in-house IT functions, there was a feeling that there was a need to assess and update the guidance. 
Since their outset the GAMP guides have promoted using a balanced risk-based pragmatism to achieve compliance and the July 2022 release of GAMP 5 second edition strongly encourages the use of critical thinking to support that.Īs ISPE GAMP 5 Second Edition notes in the preface “Operating in a highly regulated industry may lead practitioners to apply prescriptive and rigid compliance-based approaches that are not commensurate with and not effective in managing any actual risk to the product and the patient.” 1 Since 1991, GAMP ® – a community of practice within the International Society for Pharmaceutical Engineering (ISPE) – has set the tone for the validation practices necessary for regulated pharmaceutical companies to meet the computerized system expectations of the US Food and Drug Administration (FDA), the European Medicines Agency (EMA), the World Health Organization (WHO), the Medicines and Healthcare Regulatory Agency (MHRA) in the UK, the Chinese National Medicinal Products Administration and other global regulatory bodies.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
